Scientific background
IL1RAP – a Target with Several Opportunities
Cantargia develops groundbreaking antibodies targeting Interleukin-1 Receptor Accessory Protein (IL1RAP) to treat cancer and chronic inflammatory conditions. IL1RAP regulates several key disease-driving signaling pathways, and targeting IL1RAP may therefore have strong effects on development of multiple diseases.
Role of IL1RAP in inflammation
IL1RAP is a co-receptor required for signaling downstream of the IL-1, IL-33 and IL-36 receptors. When the respective cytokine binds its receptor, IL1RAP is recruited to form an active signaling complex that triggers inflammatory and immune-related pathways, mainly through NF-κB and MAPK activation. IL-1, IL-33 and IL-36 are often commonly upregulated and act together in several diseases and blockade of the individual pathways is therefore often insufficient. By targeting IL1RAP with one of Cantargia’s unique antibodies, it is possible to block all three IL-1 family cytokine pathways simultaneously to achieve superior efficacy in complex diseases.
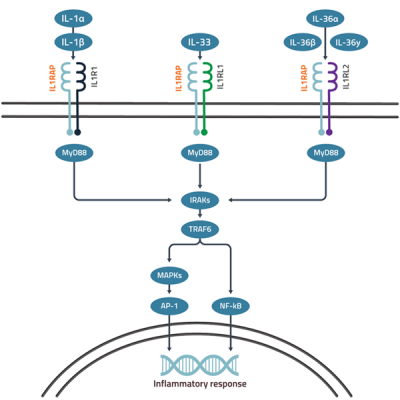
Figure 1. Schematic overview of IL-1, IL-33 and IL-36 signaling via IL1RAP.
The potent and broad benefit of IL1RAP blockade has been demonstrated in several preclinical models of inflammation, and targeting IL1RAP has proven to be more efficacious compared to single cytokine blockade (Fig. XX and refs Lema et al., Fields et al., Grönberg et al., Mulholland et al.). In addition, IL1RAP blockade has also been shown to potently reduce fibrosis (Fig. XX) extending the opportunities for therapies targeting IL1RAP from inflammation to also include fibrotic diseases.
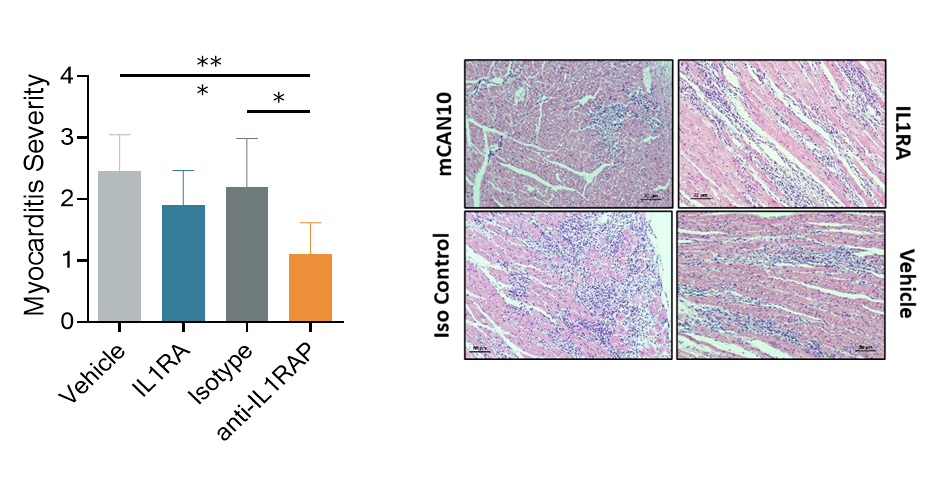
Figure 2. IL1RAP blockade reduce cardiac inflammation in a preclinical model of virus-induced myocarditis. IL1RA – blocking IL-1α and IL-1β only.
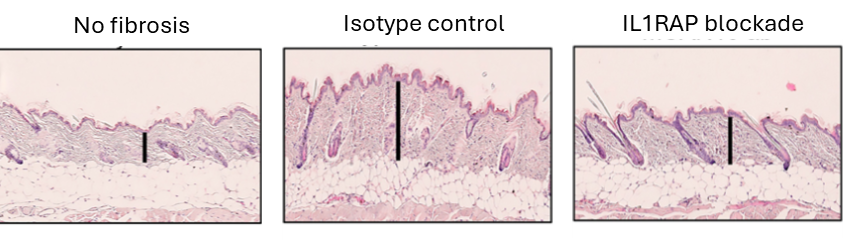
Figure 3. IL1RAP blockade reduce fibrosis in a preclinical model of skin fibrosis.
To further build on IL1RAP as a target, it could be combined with an additional functionality in a so-called bispecific antibody for stronger efficacy tailored for a specific disease. This concept is currently under investigation in the so called CAN14 project and within the CANxx platform.
IL1RAP as a key target in cancer
The key role of IL1RAP in inflammation is well established but Cantargia’s foundation is based on the discovery that IL1RAP is distinctly overexpressed across multiple tumor types. In fact, high IL1RAP expression is linked to poor outcome in patients with several types of cancer, including pancreatic cancer (Fig. XX). This indicates that blocking IL1RAP mediated signaling may be beneficial in these diseases. Also, given the high expression of IL1RAP on tumor cells, IL1RAP targeting antibodies such as nadunolimab can stimulate the body’s killer cells to find and destroy the cancer cells.
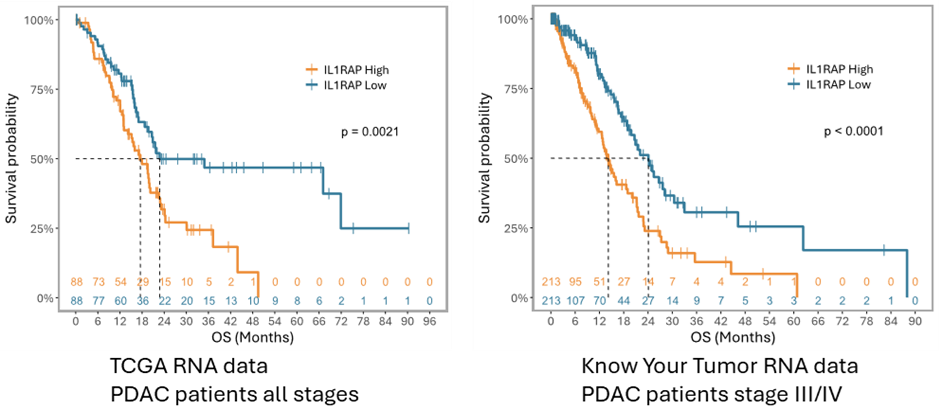
Figure 4. High IL1RAP expression is linked to poor outcome in patients with pancreatic tumors.
IL1RAP is found both on cancer cells and on several of the tumor-supporting cell types within the tumor microenvironment, including cancer-associated fibroblasts (CAFs). In this context, IL1RAP is involved in transmitting signals from mainly the two IL-1 molecules, IL-1α and IL-1β, which both contribute to a tumor microenvironment that favors tumor development and growth. For example, these signals can strengthen the tumor’s defense mechanisms against the effector immune cells exerting anti-tumorigenic functions.
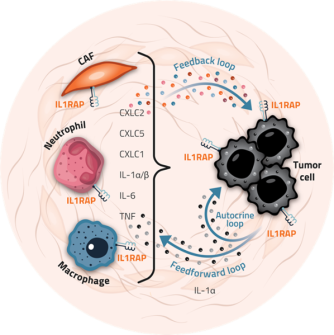
Figure 5. Schematic overview of the role of IL1RAP in the PDAC tumor microenvironment. IL-1 induced in tumor cells acts as a feedforward signaling cytokine and activates myeloid cells and CAFs in the TME. Myeloid cells and CAFs subsequently induce feedback signaling by secreting mediators that stimulate tumor cells. IL-1 family cytokines also induce collagen formation by CAFs and contributes to establishing a fibrotic, desmoplastic stroma. Activated CAFs also attract myeloid cells that fuel tumor inflammation and create an immune suppressive and treatment resistant niche.
This means that nadunolimab has a dual action – apart from inducing killing of the tumor cells it also blocks the tumor-promoting IL-1 signaling. In preclinical tumor models not responding to immunotherapy, it has been shown that IL-1 drives systemic immunosuppression and that blocking IL1RAP can restore an effective anti-tumor immune response after tumor vaccination (ref Hanahan et al).
Interestingly, the high IL1RAP expression in tumors compared to normal tissue can be utilized as a framework for delivery of payloads into a tumor microenvironment. The concept of so-called antibody-drug conjugates (ADCs) is currently being investigated under the CANxx platform.

